A team of researchers in Australia and the USA has developed a novel material that can catalyse one half of the chemical reaction needed to produce hydrogen cleanly and efficiently from nothing more than water. The reaction is one step in the light-activated oxidation, or photooxidation, of water. The material could accelerate the emergence of the hydrogen economy.
Environmentalists and technologists alike see a future in which clean energy is produced using renewable resources with minimal pollution and using hydrogen as the new currency of power. This so-called hydrogen economy will provide the fuel for powerful electric, yet clean and efficient electric vehicles and generators.

Can anyone better photosynthesis? (Credit: David Bradley)
With hydrogen being touted as one of, if not, the most important fuels of the future, the ideal production method would involve splitting water – an oxygen atom bonded to two hydrogen atoms – using an entirely renewable energy source, such as solar power, rather than converting oil-derived products using yet more fossil fuels to produce hydrogen gas by eliminating the oxygen atom and joining two hydrogen atoms from each water molecule together.
Unfortunately, the splitting of water is an energy-intensive process, which would apparently make it prohibitively expensive, but more importantly in the long term, entirely unsustainable if the electricity needed to power the electrolytic splitting of water is derived from the burning of fossil fuels. A potentially viable alternative is to use solar energy to split water.
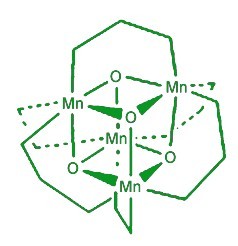
Mimicking photosynthesis (Credit: Brimblecombe et al/Angew Chem)
Now, Robin Brimblecombe and Leone Spiccia at Monash University and Gerhard Swiegers at the Commonwealth Scientific and Industrial Research Organisation in Melbourne, Australia, and Charles Dismukes of Princeton University, USA, have turned, as scientists often do, to nature for their inspiration. They have now created a catalyst using a manganese-bearing chemical modelled on the catalysts found in species that carry out photosynthesis, in which light is used to split water and ultimately form food energy by combining hydrogen with carbon dioxide from the air.
Their efforts overcome the biggest hurdle in this research field by developing a robust catalyst that can photo-oxidize water. The new catalyst is a manganese oxo complex with a cubic core comprising four manganese and four oxygen atoms capped by attendant phosphinate molecules. When light hits this chemical cube, one of the capping molecules is released to form the active species that can split water molecules.
The team has made one of the electrodes for their water-splitting system from a wafer-thin Nafion membrane on to which the catalyst is coated. This circumvents the problem that the manganese oxo cubes are not soluble in water. Housed within the aqueous channels of the Nafion membrane, the catalyst is stable and water molecules can flow past it easily. When visible light shines on the system with an applied voltage of 1.2 V, water molecules are split.
The next step will be to complement this anodic half-cell with a catalytic hydrogen-producing cathode cell. This would result in a photoelectrochemical cell that produces pure hydrogen and oxygen from water and sunlight with no applied voltage.
Further reading
Angew. Chem., 2008, in press
http://dx.doi.org/10.1002/anie.200801132
Professor Leone Spiccia
http://www.chem.monash.edu.au/staff/spiccia/
Suggested searches
catalysis
hydrogen
renewable energy
manganese