Bitter nitrogen-containing compounds found in the bark of the cinchona tree, native to South America, include the well-known antimalarial compound quinine. Quinine has been used as an additive for tonic water and is the compound that gives this drink its fluorescence. Extracts of cinchona bark have been used for centuries although the first modern usage of quinine against malaria was not until the seventeenth century.
Quinine is still in use as an antimalarial drug in some situations although the emergence of drug resistance in the malaria parasite Plasmodium falciparum meant that by the 1940s its effectiveness had declined, not coincidentally with the development of more marketable alternative drugs.
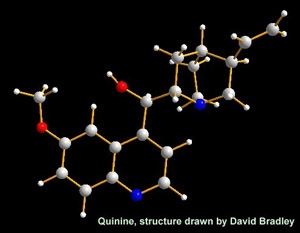
Quinine’s chemical structure (Drawn by David Bradley)
Chemists Robert Burns Woodward and William von Eggers Doering of Harvard University, first made quinine from readily available starting materials around this time, publishing details in 1945.
However, while modern chemists tend not to publish a so-called total synthesis until they have actually verified the final product, it is thought that Woodward and Doering published their formal total synthesis without having completed the final three-step reaction procedure. This procedure was developed by Paul Rabe and Karl Kindler in 1918 and perhaps the team of ’45 simply assumed it would work to give them the final quinine product, having produced the penultimate product d-quinotoxine in 1944.

Woodward and Doering recreating quinine for science photographer Fritz Goro (Credit: Fritz Goro archives)
Chemists, such as Columbia University’s Gilbert Stork, however, were less than happy with this and he dismissed the Woodward and Doering quinine total synthesis as nothing but a myth in 2001 when he himself published a formal total synthesis of the compound. Until now, no one had followed the exact procedure, other novel total synthesis schemes having precluded the need to synthesise quinine following Woodward’s recipe. In 2007, chemical historian Jeffrey Seeman traced the evidence and asserted that the Woodward Doering synthesis was a valid achievement. It was not simply a wartime propaganda myth to show that the Allies could make adequate supplies of an essential drug for their troops fighting on tropical fronts.

Robert M. Williams
Now, Aaron Smith and Robert Williams of Colorado State University have successfully reproduced the Rabe-Kindler protocol and describe their vindication of Woodward and Doering in the journal Angewandte Chemie. The article providing a fitting tribute to Doering in an issue dedicated to his 90th birthday. Smith and Williams repeated the entire procedure without resorting to any modern methods.
Indeed, following the old ways without the benefits of modern storage methods of reactive metals may have been critical in their success. Initially, their yield of quinine was very low. They suspected that the aluminium powder used as a reducing agent in the last step was the problem. It was too fresh! Leaving it in air for a short period leads to the formation of a coating of aluminium oxide. When the experiment was repeated with this powder, the yield matched that reported by Woodward and Doering.
Quinine and the cinchona bark alkaloids play an important role in modern medicine. It is thus amazing that no attempts to reproduce the Rabe-Kindler conversion of quinotoxine into quinine have been published, says Williams. Analytically pure quinine can be isolated from this reaction by the selective crystallization of the corresponding tartrate salt, just as described by Rabe in 1939, says Williams. “We have thus corroborated Rabe and Kindler’s 1918 publication. Woodward and Doering could theoretically also have followed this procedure in 1944.
Further reading
C&EN, 2007, 85, pp. 47-50
http://pubs.acs.org/cen/science/85/8509sci1.html
Robert M. Williams Research Group homepage
http://rwindigo1.chm.colostate.edu/