Could we use solar power to convert that troublesome greenhouse gas, carbon dioxide, into useful methane fuel for heating and electricity generation? According to research published in the journal Nano Letters that could be a real possibility.
The use of fossil fuels in transport and power generation has inadvertently pumped billions of tonnes of the greenhouse gas carbon dioxide into the atmosphere. Climate models suggest that the long-term impact could be to cause severe and deleterious climate change that will have an impact on us and future generations.
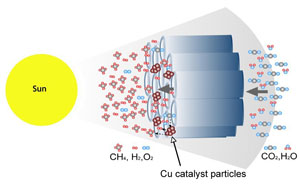
A proposed flow-through reactor for carbon dioxide conversion (Credit: Grimes Group)
Now, Craig Grimes and colleagues, Oomman Varghese, Maggie Paulose and Thomas LaTempa, at Pennsylvania State University have devised an intriguing method for converting captured carbon dioxide into useful methane, a combustible fuel, using the energy of the sun.
The team has built a material based on a three-dimensional array of nitrogen-doped titania nanotubes coated with an ultrathin layer of a platinum or copper co-catalyst. Titania normally absorbs in the high-energy ultraviolet end of the electromagnetic spectrum. However, the presence of the co-catalyst shifts this absorption into the visible wavelengths associated with sunlight reaching the Earth’s surface.

A test batch reactor for producing methane from carbon dioxide (Credit: Grimes Group)
The result is a highly efficient photocatalyst that absorbs sunlight and essentially adds this energy to carbon dioxide by converting it back into an energy-rich hydrocarbon.

Craig Grimes
To demonstrate proof of principle the team loaded their nanotube arrays into a stainless steel chamber filled with carbon dioxide and infused with water vapour. The chamber was left in direct outdoor sunlight for several hours during which time the carbon dioxide was transformed into methane and water released as a by-product. The team demonstrated that 160 microlitres of methane could be produced per hour by just one gram of nanotubes. This rate is twenty times higher than experiments carried out in the laboratory using pure ultraviolet light.
“Copper oxide and titanium dioxide are common materials,” Grimes says. “We can tune the reaction using platinum nanoparticles or ideally other, less expensive catalysts.” Grimes believes that the conversion process might readily be improved by several orders of magnitude, which could make the process economically feasible.
Capturing carbon dioxide from power stations and converting it into methane cheaply using solar power could improve the economics of carbon capture dramatically. Grimes also suggests that a similar system might also one day be possible for trapping and converting carbon dioxide from vehicle exhaust.
Further reading
Nano Lett., 2009, 9, 731-737
http://dx.doi.org/10.1021/nl803258p
Materials Research Institute homepage
http://www.mri.psu.edu
Craig Grimes spotlight
http://www.mri.psu.edu/articles/BC/faculty/CraigGrimes/index.asp
Suggested searches
greenhouse gases
carbon dioxide
methane
nanotubes